Understanding the Periodic Table
What is an Element?
An element is a pure substance made of only one type of atom.
Each element has unique properties and is represented by a symbol on the Periodic Table.
What is a Compound?
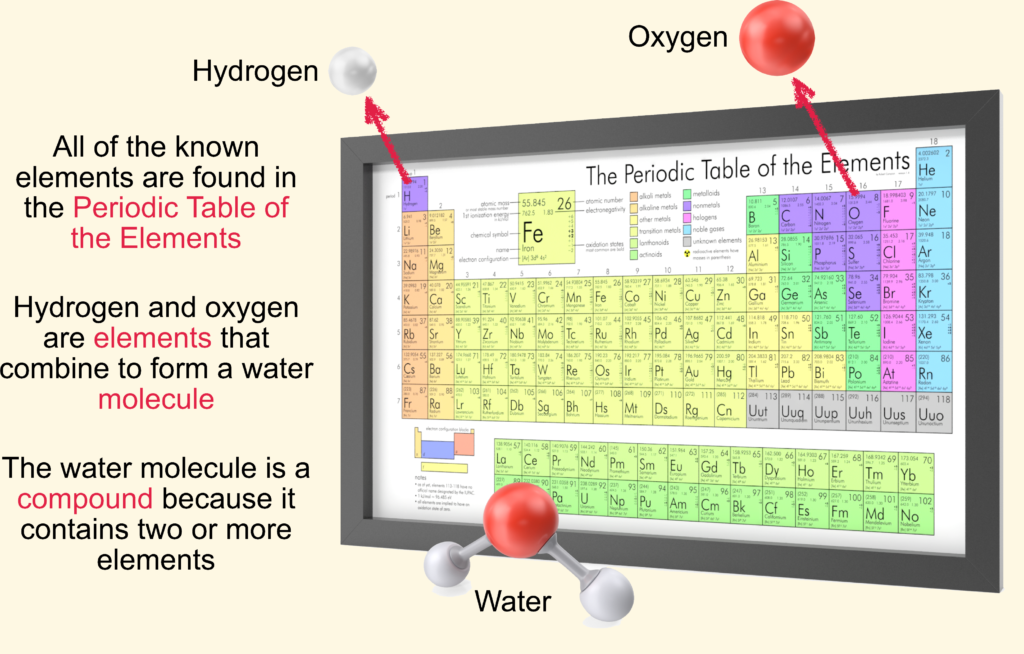
A compound is formed when two or more elements chemically combine.
For example, hydrogen and oxygen combine to form water (H₂O).
Why the Periodic Table Matters
The Periodic Table organizes all known elements based on their structure and properties.
It allows scientists to predict how substances will behave and interact.
But the Periodic Table was not simply given — it was discovered.
The Discovery of Order: Dmitri Mendeleev

Who Was Mendeleev?
In the 19th century, Russian chemist Dmitri Mendeleev developed the first widely recognized Periodic Table. He organized elements based on patterns in their properties.
A Remarkable Insight
Mendeleev noticed that when elements were arranged in a certain order, their properties repeated in a predictable way. This revealed an underlying structure in the natural world.
Predicting the Unknown
He left gaps in his table for elements that had not yet been discovered — and correctly predicted their properties.
When those elements were later found, they matched his predictions closely.
Why This Matters
The Periodic Table is not just a list — it is a powerful example of how scientific reasoning reveals order and structure in nature.
